Uveal Melanoma
Everything you need to know about uveal melanoma
Uveal melanoma is a malignant intraocular tumor that develops from pigmented cells called melanocytes, which are present within the uvea (iris, ciliary body, and choroid). It is the most common primary intraocular malignant tumor in adults.
With approximately 500 new cases diagnosed each year in France, uveal melanoma is a rare cancer for which a network certified by the French National Cancer Institute exists.
Uveal melanoma mainly affects Western populations of European descent with fair skin and light-colored eyes, usually after the age of sixty. There are no known risk factors for the majority of patients, although certain genetic predispositions exist (alterations in the BAP1 or MBD4 genes) in fewer than 2% of cases.
Due to its rarity, uveal melanoma is often difficult to diagnose. It develops insidiously long before the first symptoms appear. It may present as a pigmented spot on the iris or be visible during a fundus examination, without causing any symptoms. Conversely, it can cause vision loss, a blind spot in the visual field (scotoma), and/or repeated flashes (phosphenes) caused by retinal detachment.
Unlike conjunctival tumors, uveal melanoma cannot be confirmed by tissue biopsy. Diagnosis is therefore based on multimodal imaging performed by an ocular oncologist at a specialized center, including at minimum a photograph and an ocular ultrasound.
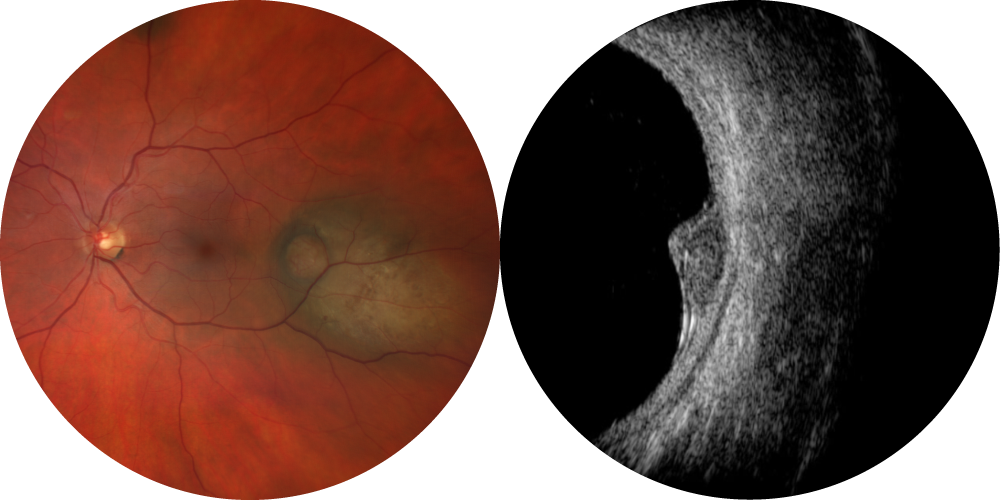
As a rare cancer, treatment must be carried out at a certified expert center (MélaChoNat) recognized by the French National Cancer Institute. The main goal of treatment is to stop the melanoma from progressing while preserving the anatomical integrity of the eye and, if possible, vision.
In most cases, uveal melanoma treatment is conservative, using dedicated radiotherapy, primarily proton therapy. Vision preservation mainly depends on the tumor volume and its location relative to the optic nerve and macula.
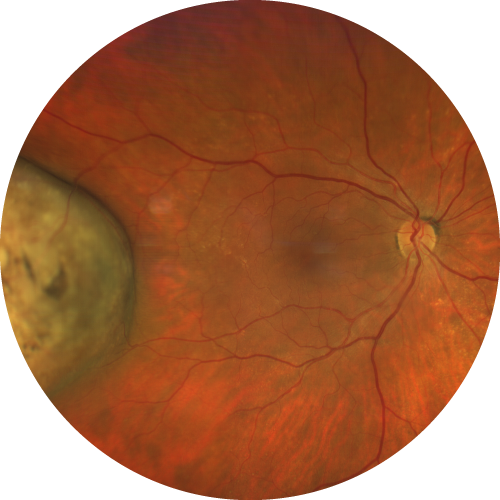

In this case, the tumor is sufficiently far from the macula and optic nerve to expect vision preservation after proton therapy.
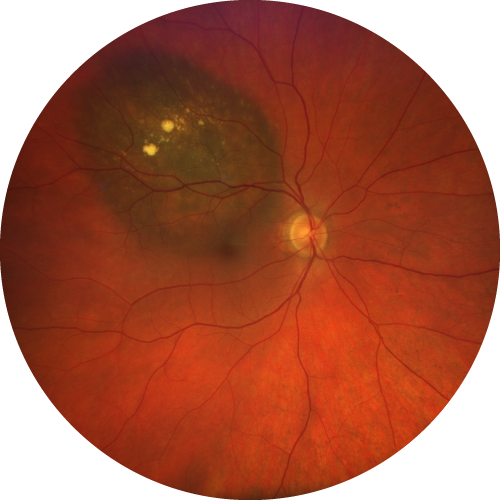

Here, the tumor is too close to the macula and optic nerve, leading to eventual vision loss after proton therapy.
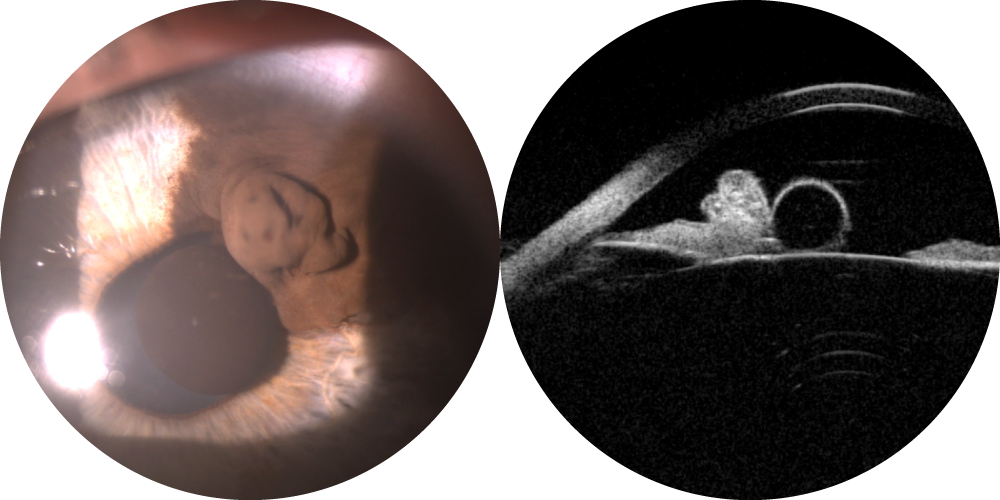
For this, marker clips are placed during specialized surgery at an expert center. Except in iris melanoma, strong light is used through the eye wall (cornea or sclera) to locate the tumor shadow, then four clips are sutured around or near the tumor. The melanoma is then treated at one of the two ocular proton therapy centers in France, in Nice or Paris.
Clips are sutured to the sclera after locating the tumor shadow by transillumination.
When the uveal melanoma is too large, in cases of local recurrence or major complications from conservative treatment, surgical eye removal (enucleation) is required. An implant is then placed with attached ocular muscles.
Ophthalmologic follow-up will be determined by the referring ocular oncologist and must be long-term, as local recurrences can occur even after five years from initial treatment. Conservative treatment is effective in the majority of cases (approximately 95% after more than 10 years). There is no risk of uveal melanoma spreading to the other eye in the absence of metastasis.
Systemically, the occurrence of metastases is unfortunately common, in about half of cases. Metastases usually develop in the liver, requiring long-term follow-up with non-radiating liver imaging (ultrasound or MRI) for 10 years. Risk depends mainly on initial tumor volume, ciliary body involvement, and tumor genomic features (notably on chromosomes 3 and 8). Eye treatment, whether conservative or not, does not change this risk. Metastatic treatment is limited to a few systemic or regional therapies. A specific immunotherapy for uveal melanoma metastases (tebentafusp) is currently available in France for patients with the HLA*A02:01 antigen (about 27% of white European populations – doi:10.1016/j.humimm.2013.06.025).
Research efforts are focused on biological risk analysis for uveal melanoma metastasis, as well as on the type of sample analyzed. Some clinical trials are available in France at a few expert centers, testing non-destructive treatments or adjuvant therapies before and/or after treatment of localized ocular melanoma. New proton therapy modalities (FLASH) are under study. Clinical trials are also available for metastatic relapse at Institut Curie and Centre Antoine Lacassagne.